Drug Safety
How safe and effective are new therapies in clinical routine?
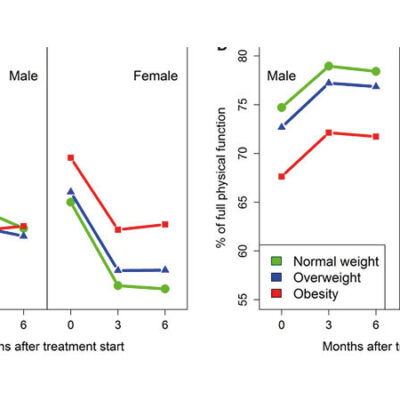
If new drugs are approved, their safety and efficacy in everyday life still have to be proven. Patients in routine care suffer more frequently from other diseases, are older and often more limited in their physical function than patients included in clinical studies. In particular, the long-term safety of the therapies sometimes used throughout their life cannot be assessed at the time of approval due to the short duration of clinical studies.
Many questions a doctor asks himself when prescribing new therapies remain unanswered.
The Research group Pharmacoepidemiology wants to close many of these knowledge gaps in rheumatology with the results of its observational cohorts.
RABBIT
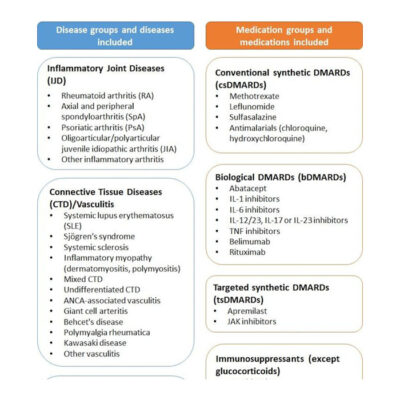
The most important register to date is the long-term cohort study RABBIT. Since 2001, this observational study has included patients starting a new therapy (either with a biologic, biosimilar or JAK inhibitor) or, as a control group, patients switching from one conventional therapy to another. More than 17.000 patients with rheumatoid arthritis have already been included. Their data is regularly documented for at least 5 and often even 10 years by about 300 rheumatologists all over Germany.
RABBIT is one of the world’s largest registers of biologicals.
Results from RABBIT
- In cooperation with other national and international partners, we carry out analyses such as the occurrence of malignant melanoma. Thus, we were able to invalidate a previously reported risk increase under therapy with TNF inhibitors.
- The results of an analysis of the incidence of lymphoma under biologic therapy are also encouraging: with almost 600.000 patient-years of observation, the risk was not increased compared to conventional standard therapy.
- However, another risk is confirmed by the data from RABBIT: In connection with the administration of biologics, the risk of suffering a serious infection increases by about 80 percent compared to conventional therapy. However, it must be taken into account that both a strong disease activity and an often additionally administered high dose of glucocorticoids also increase the risk of infection. However, as soon as disease activity is successfully reduced – for example by biologic therapy – and the glucocorticoid dose is reduced and the patient’s physical function is improved, the risk of infection decreases again.
- The risk of sepsis or death after a serious infection is significantly reduced in patients who received a biologic agent at the time of infection.
RABBIT data dispelled the suspicion that TNF inhibitors promote the development of heart failure. Rather, we were able to show that the use of these biologics can prevent both the worsening of existing heart failure and the occurrence of new heart failure. - With regard to myocardial infarction, we showed that not biologic therapy, but above all the high inflammatory activity of the rheumatic disease increases the risk of the event.
- This also applies to the occurrence of a stroke.
RABBIT Risk Score
The RABBIT risk score estimates the probability of a patient with rheumatoid arthritis to suffer from a serious infection during the next 12 months. This is done on the basis of information on age, comorbidities, and previous and current therapy.
The score is freely accessible.
RABBIT-SpA
Since 2017, data from patients with axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) have been collected in this web-based monitoring register, comparable to RABBIT, in order to investigate the efficacy and safety of the therapies used in the long-term.
Rhekiss - Rheumatism, fertility and pregnancy
The Rhekiss Register was started in 2015 and monitors the course and outcome of pregnancies until the child’s second birthday in patients with various inflammatory rheumatic diseases who receive different therapies. This information is intended to increase safety in the care of pregnant women with rheumatic diseases and to improve the counselling of patients wishing to become pregnant and during pregnancy.
950 patients have already been entered in the register, 207 of them in the module for women with wish to have a child.
Data Collection 2.0
In contrast to the paper-based RABBIT register, RABBIT-SpA and Rhekiss are web-based registers. Patients complete their questionnaires via tablet, computer or smartphone app; medical documentation by rheumatologists is done on the computer.

 Deutsch
Deutsch